Most existing methods report γ-C( sp 3)–H functionalization at the terminal methyl sites (Fig. However, such reverse site selectivity is yet to be explored in the more distant sites. In this context, β-methylene C–H activation in the presence of methyl has been explored recently (Fig. To push the boundary further, we envisaged if weak coordination of carboxylate could induce challenging methylene activation in the presence of the equally accessible methyl group. 
However, C–H activation was carried out mostly at the terminal methyl group. Recent years have witnessed a great progress in this domain where a number of transformations have been achieved at the proximal and distal sites of aliphatic carboxylic acids 12, 13, 14, 15, 16. Exploiting the weak coordination of the carboxylate group for challenging activation of C( sp 3)–H bonds offers great advantage as it sidesteps the necessity of using an external directing group. Alkyl carboxylic acids have come up as one of the most promising substrates to transform into valuable entities via Pd-catalysed C( sp 3)–H activation. In this Article, we sought to develop a simpler and more effective method to access bicyclic lactones that could overcome the existing limitations. However, the weak coordination of the carboxylate group has been shown to activate mostly the methyl group at the β or γ positions, limiting the scope and applicability of aliphatic acids 8, 9, 10, 11. In this context, aliphatic acids have been identified as the choice of substrates that could be functionalized without utilizing any extra directing group. Nonetheless, selective transformation of distal aliphatic C–H bonds still presents an uphill challenge, more so in the absence of an exogenous directing group 7 that is added externally to the substrate. Transition metal catalysis has become a powerful tool to convert unreactive aliphatic C–H bonds into an array of useful functionalities 4, 5, 6. The past two decades witnessed a notable advance in synthetic organic chemistry, where unactivated C–H bonds were selectively transformed into complex organic molecules. Nonetheless, traditional synthetic approaches suffer from atom and step economy due to the complex starting material preparation, multi-step procedures, unwanted side product formations and so on. Numerous efforts have been made to prepare bicyclic lactones using different synthetic strategies 3. Constructing these cyclic architectures has been a topic of great interest among synthetic chemists not just because of their abundance in nature, but also because of their utility as versatile synthons (Fig. The method substantially simplifies the synthesis of important bicyclic lactones that are important features of natural products as well as pharmacoactive molecules.īicyclic lactones comprise a large number of natural products with diverse biological activities (Fig. 
The broad generality of this reaction has been highlighted via dehydrogenative lactonization of mid to macro ring containing acids along with the C–H olefination reaction with olefin and allyl alcohol. Computational studies suggest that reversible C–H activation is followed by β-hydride elimination to generate the Pd-coordinated cycloalkene that undergoes stereoselective C–O cyclization, and subsequent β-hydride elimination to provide bicyclic unsaturated lactones. The protocol allows the reversal of the general selectivity in aliphatic C–H bond activation. 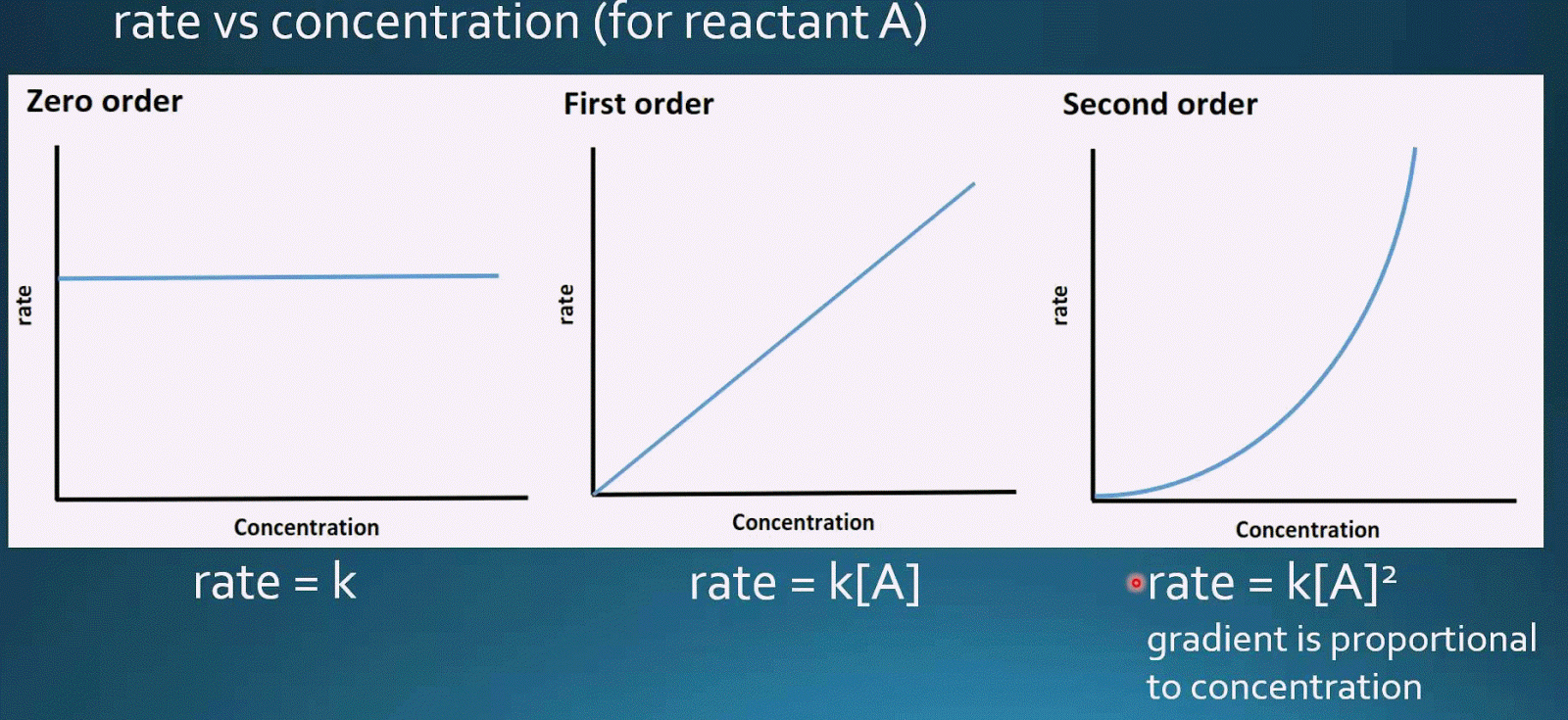
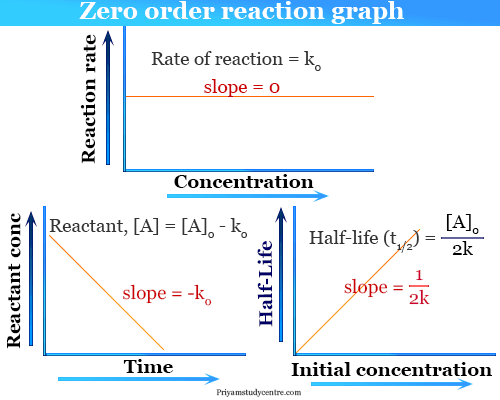
Here we show activation of a methylene C–H bond in the presence of methyl C–H bonds to form unsaturated bicyclic lactones. Activation of a methylene C–H bond in the presence of methyl C–H is underexplored. However, regioselective activation of distal aliphatic C–H bonds poses a tremendous challenge, particularly in the absence of directing templates. Unlike with first-order reactions, the rate constant of a second-order reaction cannot be calculated directly from the half-life unless the initial concentration is known.Transition metal catalysis plays a pivotal role in transforming unreactive C–H bonds. Consequently, we find the use of the half-life concept to be more complex for second-order reactions than for first-order reactions. We can derive the equation for calculating the half-life of a second order as follows: $$\frac $ is inversely proportional to the concentration of the reactant, and the half-life increases as the reaction proceeds because the concentration of reactant decreases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
